The French version of this article is still available here
Ammonia is the second most produced molecule (in terms of annual volume) by the chemical industries. This key molecule, with major applications in the agricultural sector, also has the ability to store energy (in particular hydrogen). Moreover, ammonia in liquid form can also be used as a fuel to replace LPG of fossil origin.
As it is not carbonaceous, ammonia used as a fuel does not emit CO2 in its use phase. For this reason, it appears as a strategic solution in the maritime and air transport sector as a replacement for heavy fuel oils and kerosene respectively.
If these arguments seem so beneficial and indisputable, various questions deserve to be raised. At present, ammonia is produced from fossil resources. Is it therefore technically possible to produce this molecule from renewable resources? Is ammonia really the « fuel of the future » as some manufacturers advocate?
Are we moving towards an « ammonia economy »? That’s the question I’m answering today.
Prelude
Ammonia is a molecule with the chemical formula NH3 that occurs as a gas at room temperature and normal pressure. It can also be stored as a liquid at low temperatures (below -33°C) and/or when compressed. In this case, it is called liquid ammonia.
Ammonia is a base, irritating both to the skin and to the respiratory tract, which has a specific odor detectable in the air at doses of only a few ppm. This gas is highly soluble in water with solubilities that decrease with increasing temperature (the solubility in water goes from 89.9 g NH3/100 g water at 0°C to 7.4 g NH3/100 g water at 96°C). When ammonia is solubilized in water, we have a solution of aqueous ammonia (NH4OH).
Ammonia takes its name from the god Ammon and more specifically from the pieces of « salammoniac » (also called « salt » ammonia), a mineral with a typical odor and composed of ammonium chloride (NH4Cl) collected by the Romans near the temple of Jupiter-Ammon in ancient Libya. Since antiquity, men were aware of the existence of this molecule, and its « olfactory » characteristics. An emblematic molecule of certain alchemical approaches, it was not until 1785 and the conclusions of the French chemist Berthollet before the Academy of Sciences that the centesimal composition of this molecule was understood and it was established that it was composed of a nitrogen atom on which three hydrogen atoms were fixed.[1] Knowledge about ammonia will also gradually expand towards the middle of the 19th century with the discovery and description of the nitrogen cycle. It was at this time that ammonia was revealed as a constituent of choice for the formulation of nitrogen fertilizers and that the question of large-scale production of this molecule began to be raised.
Natural abundance and industrial synthesis
Ammonia is ubiquitous in the atmospheres and soils of many planets (and satellites) of our solar system, whether in gaseous or solid form.[2] A study published in August 2020 describes the presence on Jupiter of violent storms generating hailstones of ammonia.[3] On Earth, on the other hand, it is only detected in trace amounts, mostly from animal or plant matter or by anthropogenic emissions via the industrial sector. However, ammonia is found in the deep layers of the earth, trapped as salts, as well as in certain rocks or fossil organic matter.[4] Ammonia is also detected in rainfall, some volcanic regions, as well as in fertile soils.
Until the 19th century, ammonia was produced by distillation of manure or by extraction of domestic black water. It was then obtained after the second half of the 19th century as a by-product of the manufactured gas industry (town gas).[5] It was not until the beginning of the 20th century (1909-1913) that the Haber-Bosch process, one of the most efficient processes in the chemical industry (all categories combined), was developed and is still used today.
The Haber-Bosch process, which is of considerable economic significance, quantitatively combines two gases (hydrogen H2 and nitrogen N2) typically at pressures between 100 and 300 bar, and temperatures in the range of 300-550°C in the presence of an iron (Fe3O4) catalyst (Figure 1). The reaction is exothermic, which means that it releases energy in the form of heat. At the end of the process, the ammonia is separated from H2 and N2 (which are re-injected into the process) by liquefaction and stored.
![]()
Nitrogen (N2), one of the inputs to this process, is catalytically fixed from atmospheric air. As for hydrogen (H2), it is obtained upstream from catalytic reforming of natural gas (methane).
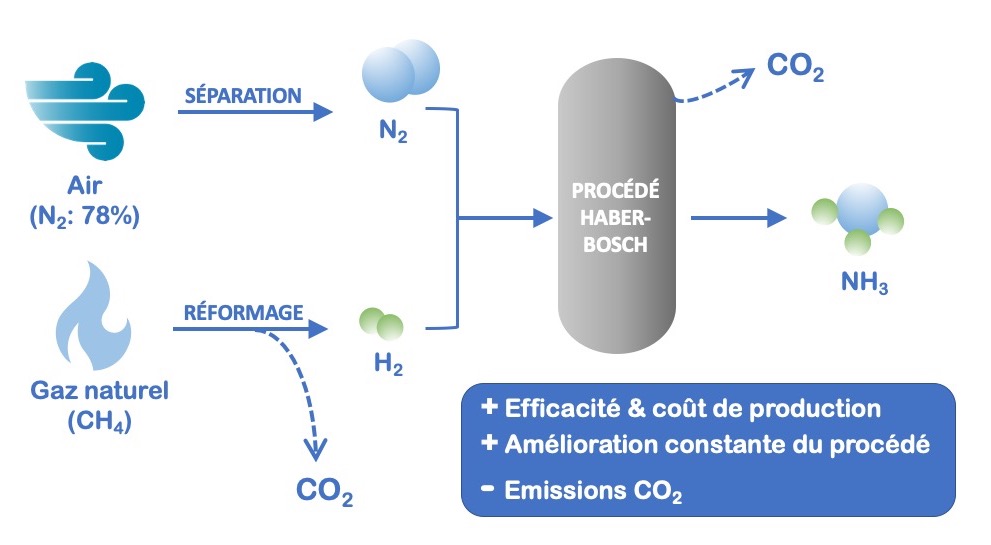
Figure 1: Haber-Bosch process diagram
According to the Institute for Industrial Productivity’s Industrial Efficiency Technology Database, industrial ammonia production peaked at more than 164 million tons in 2011, corresponding to a daily output of nearly 450,000 tons. Production has been increasing for more than 20 years, with annual volumes rising by 25% between 2000 and 2010. The biggest increase is noted in China over the last 15 years (+65%) as well as in countries like India, Russia, or even Trinidad and Tobago. The ammonia market is fragmented with different players occupying the market share. The market is expected to grow at around +2% for the period 2019-2023.[6]
Between 75-90% of annual ammonia production is for the fertilizer sector (it is estimated that more than 50% of food production relies on ammonia-based fertilizers).[7] The remaining 10-25% of ammonia production is used in the pharmaceutical, textile, fine chemical and plastics industries.
Although the Haber-Bosch process is one of the most efficient and effective processes in the chemical industry, it should be mentioned that it consumes (due to the high pressures and temperatures required for the reaction) almost 1% of the world’s energy production! This process would thus represent more than 17% of the energy consumed by the chemical and petrochemical sectors. In 2004, it was established that the production of NH3 used more than 5.6 EJ of fossil fuels, of which 2.9 EJ were for the natural gas used to produce H2 and 2.7 EJ for the synthesis process itself. Moreover, the overall carbon footprint of the process is unfavorable, with concomitant CO2 emissions (the generation of H2 by reforming causes more than 50% of the carbon emissions of the entire ammonia production chain). It is estimated that for each molecule of NH3 produced, one molecule of CO2 is emitted on the whole production chain.
Towards an industrial production of ammonia with a smaller carbon footprint?
Aware of the ever-increasing projections of ammonia demand, and the environmental issues associated with its industrial production, researchers (academic and industrial) are focusing on continuous process improvements and more innovative production routes.
The use of renewable resources as inputs in the production schemes could offer a solution in the short or medium term, for example by exploiting water electrolysis as an H2 production process. The use of solar energy (photovoltaic panels) alone could reduce the CO2 emissions associated with the production of NH3 by more than 50% in the Haber-Bosch process (Figure 2). A pilot plant of this type is currently being evaluated by Yara in Australia.
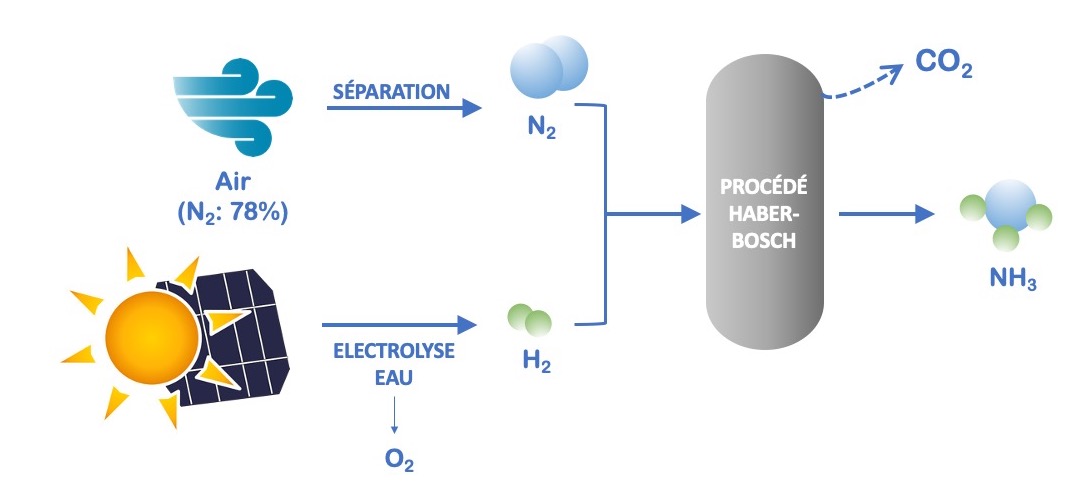
Figure 2. Improving the ammonia production process by using renewable energy sources.
Other disruptive approaches are also being studied, including electrolysis and low-pressure, low-temperature combination of H2 and N2, powered by solar and wind energy (Figure 3).
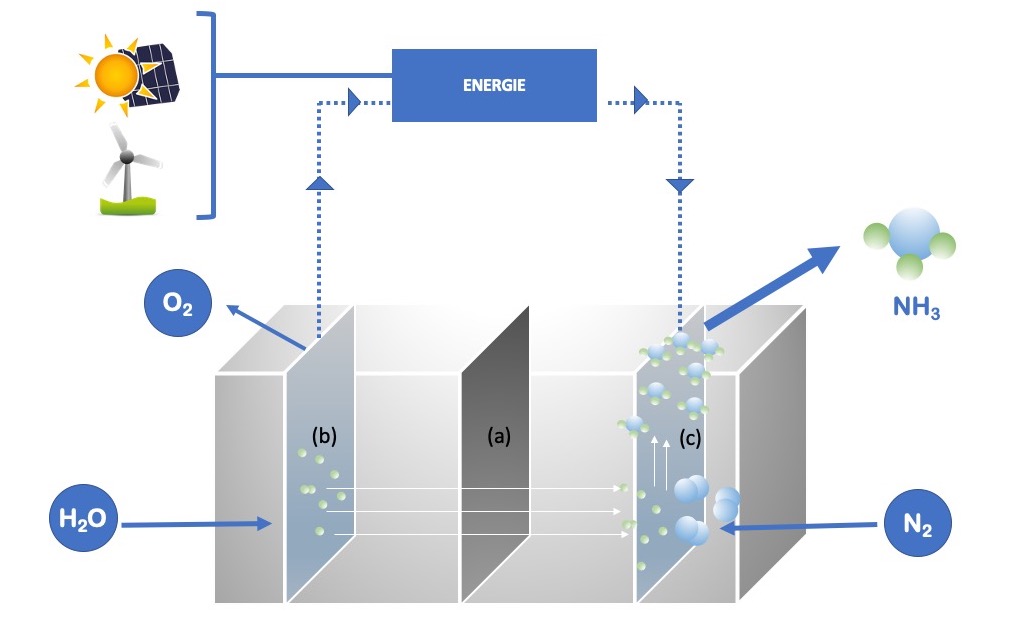
Figure 3. Alternative ammonia production at low T° and pressure in an electrolysis unit powered by renewable energy. (a) = separation membrane; (b) = anode; (c) = cathode. The dotted arrows indicate the path of the electrons within the device.[8]
It should be noted, however, that plants manufacturing ammonia take advantage of the concomitant carbon dioxide emissions that they exploit to produce urea (CO(NH2)2), a molecule whose annual production amounts to more than 120 million tons. This is called integration, which means combining or taking advantage of chemical production on the same site.
Urea is synthesized by a reaction between ammonia and CO2 at high pressure (Figure 4). Urea is mainly used in agriculture as a fertilizer and as a feed additive.
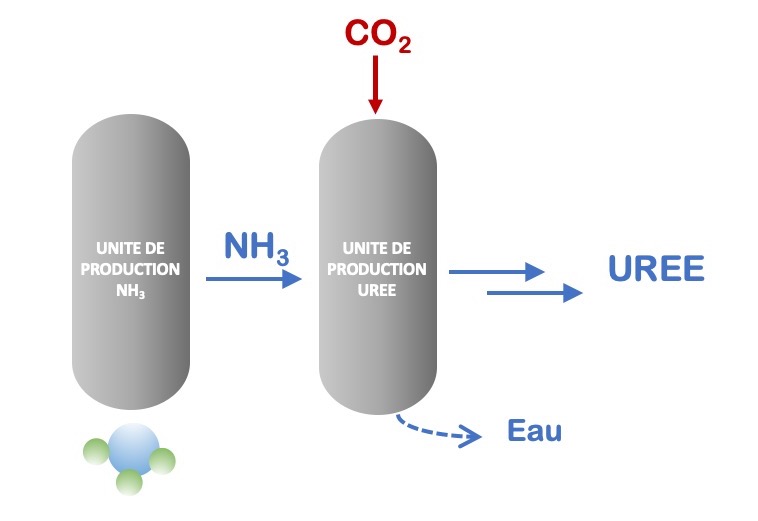
Figure 4: Summary illustration of integrated urea production
Liquid ammonia: competitive storage and usage properties?
With efficient industrial production, which is constantly improving, and with a growing emphasis on the use of renewable resources to reduce the carbon footprint of the process, ammonia is a molecule that is increasingly emerging as a strategic issue in the energy sector, either directly liquefied for use as a fuel in internal combustion engines, or as a carrier for hydrogen storage.[9]
Liquid ammonia can thus be used as a fuel for transportation but also as a fuel for heating (domestic or industrial). Ammonia can be fractionated to generate hydrogen for use in fuel cells and other targeted applications related to transportation or heat and power generation.
The combustion of ammonia generates only water and nitrogen (N2) (and heat).
4 NH3 + 3 O2 → 2 N2 + 6 H2O + Heat
Thus, ammonia does not produce any carbon emissions (CO2) as is usually the case with other liquid fossil fuels (hydrocarbons). Nitrogen oxide (NOx) emissions are reported under certain conditions of use, but this information is still incomplete and needs to be better documented and anticipated.
Liquid ammonia has a volume energy density of 11.5 MJ/L, which is about three times less than diesel, or half that of ethanol (a biofuel added to gasoline) (Figure 5). Liquid ammonia is thus more compact than some other « traditional » fuels such as gasoline or kerosene or some « biofuels » such as ethanol or butanol. It is also easily stored in liquid form (much more than hydrogen). Its boiling temperature (bp) and its condensation pressure (Pc) are close to those of propane (used as LPG – Liquefied Petroleum Gas – in transport) (bp NH3 = -33.4°C at 1 atm against -42.1°C for propane; Pc NH3 at 25°C = 9.9 atm against 9.4 for propane) This analogy would therefore make it possible to use, for liquid ammonia, the transport and storage infrastructures already in place for LPG (transport ships, storage tanks, pipelines, distribution structures).
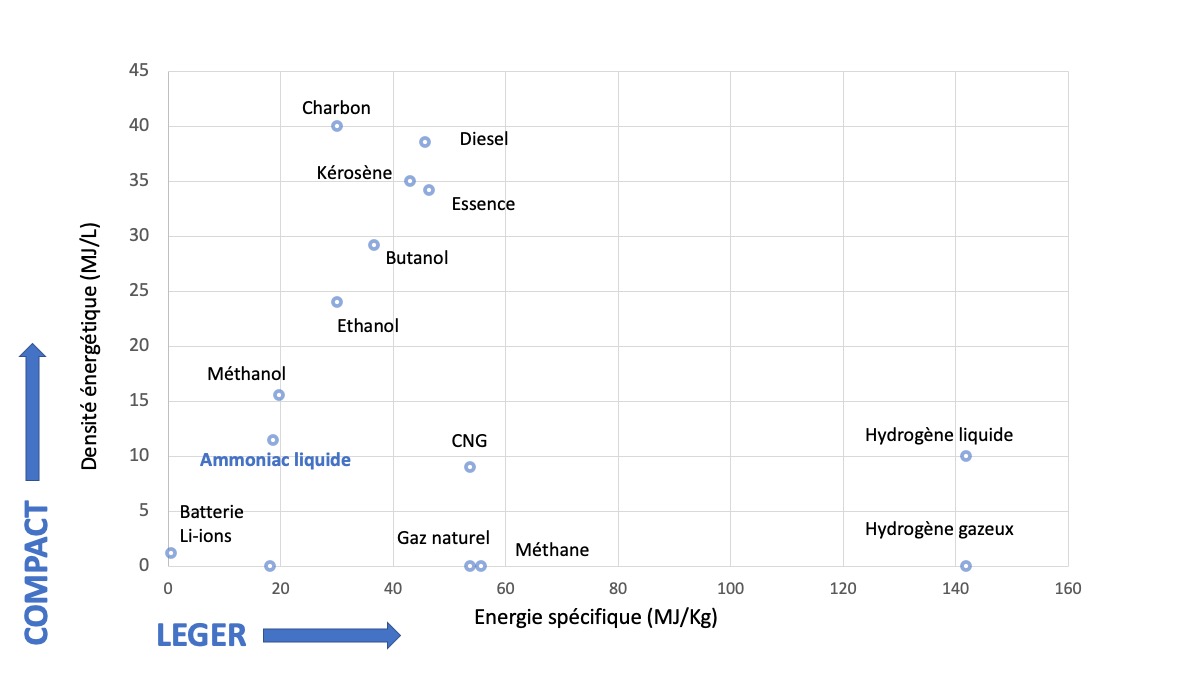
Figure 5. Energy density of some products (solid, liquid or gas).
However, ammonia is more toxic than other conventional fuels. However, it is less flammable, and therefore presents less of an explosion hazard than compressed natural gas (CNG), methanol, gasoline, hydrogen or LPG (Figure 6).
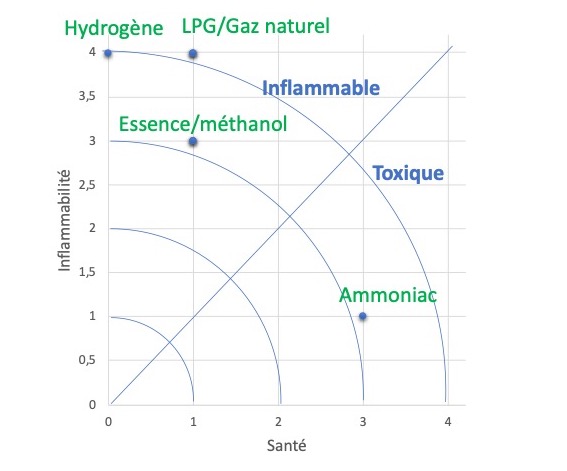
Figure 6. Toxicity vs. flammability of various fuels. 0 = no risk, 4 = severe risk.
Use « as is » in combustion engines
The use of ammonia as fuel is not recent. It was already used since the beginning of the 19th century as a fuel in motorized vehicles, in particular in locomotives (England) or in tramways (New Orleans, USA). During the Second World War, Belgium (then a victim of an embargo on diesel) even decided to power its buses with liquid ammonia. In the 1960s, ammonia was also considered for use in alternative engines, especially for military purposes. Ammonia was used as a propellant for some rocket planes in the 1950s and 1960s in a series of suborbital missions.
More recently, ammonia has come back to the forefront in order to « decarbonize » various specific sectors, mainly the maritime transport sector as a replacement for certain heavy fuels (HFO type), known for their greenhouse gas emissions.[10] Ammonia is less risky than hydrogen during storage operations and emits less greenhouse gases than LPG or CNG. It is therefore considered an economically viable fuel for the maritime transport sector. However, its overall environmental impact (from production to use) must be assessed on a case-by-case basis. Indeed, the transport of ammonia from its production area to the port areas must be as short as possible (in terms of distance). Various initiatives to demonstrate the potential of ammonia in the maritime sector are gradually being set up, particularly in the Netherlands.[11]
The efficiency of ammonia in internal combustion engines is improved when it is blended with other fuels. Ammonia has a low flame speed and high resistance to self-ignition. Doping ammonia with other fossil fuels (especially diesel) is the most technically efficient option, reducing CO2 and NOx emissions if the NH3 content of the blend does not exceed 60% by weight. Gasoline/NH3 or ethanol/NH3 blends also offer high power output under stable conditions, although mainly conditioned by NOx emissions during the combustion phases.
An energy carrier for hydrogen storage
Ammonia can also be used to store hydrogen. The hydrogen mass percentage in ammonia is quite high (17.6%) compared to other storage solutions such as methanol (12.5%).[12] However, to produce hydrogen (H2) from ammonia, through cracking operations, a significant external energy input is required, as well as very large capacity reactors (or tanks on board vehicles). The by-product of NH3 decomposition into hydrogen is nitrogen (N2) which can be emitted into the atmosphere without any environmental risk.
![]()
The ammonia decomposition reaction is endothermic, which means that large temperature inputs (> 600°C) are required, in the presence of a catalyst (often nickel-based). These temperatures are well above the operating temperatures of proton exchange membrane fuel cells. The % of unconverted NH3 at 1 bar goes from 0.88% at 400°C to 0.10% at 600°C and becomes negligible around 900°C (0.015%). The current fuel cell membranes are sensitive to residual traces of ammonia, so it is logical to favor the highest possible decomposition temperatures. The choice of materials for the manufacture of on-board tanks is therefore crucial, allowing them to withstand the high variations in T° applied (from ambient T° when the vehicle is not in use to the T° required for the production of hydrogen). In addition, the material must be able to withstand pressure variations, as well as being corrosion resistant.
The use of ammonia as a hydrogen carrier in on-board tanks is thus subject to certain constraints (technical and safety) as well as to technological barriers that still need to be overcome by researchers. It is thus important to increase the safety aspects related to the use of ammonia (remember that ammonia is toxic). In addition, the design of new catalysts to improve the conversion of ammonia into hydrogen at lower temperatures must be studied.
Conclusion: are we heading towards an « ammonia economy »?
Ammonia is a strong production of the chemical sector. However, its production cost (in euros/ton) is dependent on the price of natural gas (raw material).[13] In a simplified and prospective way, for a natural gas price rising from 1.5 USD/million BTU (February-March 2020 price) to 4.1 USD/million tons BTU (November 2018), the price of ammonia thus oscillates between 25 USD/ton and 120 USD/ton.[14] An increase in the price of natural gas to 10.5 USD/million BTU would result in an ammonia price of 377 USD/ton, which would provide a market price of H2 at over 3 USD/kg (assuming 75% efficiency for NH3-H2 cracking). The value of transitioning to renewable resources as inputs into production schemes and/or as a source of energy is therefore a considerable strategic issue in the deployment of ammonia as the molecule of choice in the energy sector.
Japan was one of the first countries to look at ammonia as a hydrogen storage medium.[15] The « Green Ammonia » program, bringing together a consortium of academics and industries including Tokyo Gas, and financed by public funds, aims to build a low CO2 emission ammonia value chain for fuel & power applications.
In the United States, the Renewable Energy to Fuels through Utilisation of Energy-Dense Liquids (REFUEL) program, funded by the Department of Energy, aims to convert electrical energy from renewable resources into energy-dense liquid fuels (including ammonia) that can, on demand, deliver electricity or hydrogen. England is also interested in ammonia (in partnership with Siemens) and more specifically in the study of how wind energy can be converted into ammonia for storage and later returned as energy via an internal combustion engine.
In Australia, ammonia is also being intensively investigated for energy storage and production. In particular, efforts are being made to reduce the carbon footprint of the ammonia production process via the Haber-Bosch route (Figure 2) or by promoting the development of electrolysis techniques (Figure 3).
In Europe, the Netherlands is leading the way in promoting ammonia due to the large amounts of renewable resources available as a result of private and public investments in recent years. Ammonia thus appears as an energy carrier for the domestic market but also can be exported. Companies such as NUON, Gasunie, Statoil and OCI Nitrogen are currently evaluating the conversion of one of the three 440 MW gasifiers at the Magnum Power plant to use hydrogen, and possibly ammonia, in super batteries that will power the plant by 2023-2030.
Maritime players are also exploring the option of using ammonia as a fuel for merchant ships with improved storage options in large ship containers that not only mobilize ammonia, but can also use it for refueling purposes. C-Job Naval and Proton Venture are part of a consortium seeking funding opportunities to develop a new generation of ammonia-capable super-ships by 2040.
Finally, the International Energy Agency (IEA), which is interested in the use of ammonia for power generation globally, has recently become one of the most active proponents of ammonia, and its production from renewable resources (wind, solar, tidal, etc.). A recent IEA study shows that ammonia is much cheaper to store over a long period of time than hydrogen, $0.5/kg-H2 for ammonia versus $15/kg-H2 for hydrogen over a six-month period, and at least three times cheaper to ship offshore or onshore.9
Ammonia research must therefore be deepened, substantiated and argued in order to position itself even more assertively at the forefront of the international energy scene.
Would you like more information on this subject?
Do not hesitate to contact me via the following email address: a.richel@uliege.be or via the form available by clicking here.
Notes and references
[1] Lemay, Pierre. Berthollet découvre la composition de l’ammoniac. Dans : Revue d’histoire de la pharmacie, 48ème année, n°165, 1960, pp. 346-348.
[2] Brown, M. E.; Calvin, W. M. Evidence for Crystalline Water and Ammonia Ices on Pluto’s Satellite Charon. Science (80). 2000, 287 (5450), 107–109. https://doi.org/10.1126/science.287.5450.107. https://science.sciencemag.org/content/287/5450/107.abstract
[3] Becker, H.N., Alexander, J.W., Atreya, S.K. et al. Small lightning flashes from shallow electrical storms on Jupiter. Nature 584, 55–58 (2020). https://doi.org/10.1038/s41586-020-2532-1
https://www.nature.com/articles/s41586-020-2532-1#citeas
[4] https://agupubs.onlinelibrary.wiley.com/doi/10.1029/2002GB001862
[5] https://www.societechimiquedefrance.fr/Ammoniac.html
[6] http://www.iipinetwork.org/wp-content/Ietd/content/ammonia.html#key-data
[7] https://cen.acs.org/environment/green-chemistry/Industrial-ammonia-production-emits-CO2/97/i24
[8] Giddey, S., Badwal, S. P. S., Kulkarni, A. Review of electrochemical ammonia production technologies and materials. Int. J. Hydrogen Energy 38 (2013), 14576-14594.
[9] Valera-Medina, A., Xiao, H., Owen-Jones, M., David, W.I.F., Bowen, P. J. Ammonia for power. Prog. Energy and Combustion Sci. 69 (2018) 63-102.
[10] McKinlay, C. J., Turnock, S. R., Hudson, D.A. A comparison of hydrogen and ammonia for future long distance shipping fuels. LNG/LPG and Alternative Fuels, 29-30 janvier 2020, Londres
[11] https://www.rivieramm.com/news-content-hub/using-ammonia-as-a-fuel-for-tankers-55211
[12] https://www.frontiersin.org/articles/10.3389/fenrg.2014.00035/full
[13] https://www.energy.gov/sites/prod/files/2015/01/f19/fcto_nh3_h2_storage_white_paper_2006.pdf
[14] https://www.indexmundi.com/fr/matieres-premieres/?marchandise=gaz-naturel&mois=60
[15] https://greenammonia.org/index_eng.html



